| IODP Proceedings Volume contents Search | ||

|
||
| Expedition reports Research results Supplementary material Drilling maps Expedition bibliography | ||
|
doi:10.2204/iodp.proc.311.209.2008 IntroductionMicrobial sulfate reduction is the major pathway of organic matter oxidation in coastal marine and continental shelf sediments (Jørgensen, 1982) and is a fundamental process linking the geochemical cycles of carbon, sulfur, and oxygen (e.g., Schidlowski et al., 1983; Berner, 1982; Garrels and Lerman, 1984; Wortmann and Chernyavsky, 2007). Sulfate-reducing microorganisms reduce SO42– according to the following net reaction:
SO42– + 2CH2O
Microbially mediated sulfate reduction affects the isotopic composition of dissolved and solid sulfur species in marine sediments. Although several details of the fractionation process remain controversial, the overall process is well understood and can be described as the sum of several mass-dependent fractionations during the stepwise reduction of sulfate to sulfide and the ratio between the forward and backward reactions (Rees, 1973; Bruechert, 2004; Brunner and Bernasconi, 2005). Experiments and field data show that the
This data report offers |
|

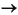 H2S + 2HCO3–.
H2S + 2HCO3–.
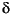 18OSO4 composition is also modified in the presence of sulfate-reducing microorganisms (Mizutani and Rafter, 1973; Böttcher et al., 1998; Brunner et al., 2005). This has been attributed either to a kinetic isotope effect during the reduction of sulfate to sulfite or cell-internal exchange reactions between enzymatically activated sulfate (adenosine phosphosulfate [APS]) and/or sulfite with cytoplasmic water (Mizutani and Rafter, 1973; Fritz et al., 1989; Wortmann et al., 2007) and/or between sulfite and adenosine monophosphate during APS formation. The isotopic fingerprint of these processes may be further modified by the cell-external reoxidation of sulfide to elemental sulfur and the subsequent disproportionation to sulfide and sulfate or by the oxidation of sulfite to sulfate.
18OSO4 composition is also modified in the presence of sulfate-reducing microorganisms (Mizutani and Rafter, 1973; Böttcher et al., 1998; Brunner et al., 2005). This has been attributed either to a kinetic isotope effect during the reduction of sulfate to sulfite or cell-internal exchange reactions between enzymatically activated sulfate (adenosine phosphosulfate [APS]) and/or sulfite with cytoplasmic water (Mizutani and Rafter, 1973; Fritz et al., 1989; Wortmann et al., 2007) and/or between sulfite and adenosine monophosphate during APS formation. The isotopic fingerprint of these processes may be further modified by the cell-external reoxidation of sulfide to elemental sulfur and the subsequent disproportionation to sulfide and sulfate or by the oxidation of sulfite to sulfate.